Stacey, Nuclear Reactor Physics, John Wiley & Sons, 2001, ISBN: 0- 471-39127-1. Baratta, Introduction to Nuclear Engineering, 3d ed., Prentice-Hall, 2001, ISBN: 8-1. Lamarsh, Introduction to Nuclear Reactor Theory, 2nd ed., Addison-Wesley, Reading, MA (1983). This defines an electron shell, which is the set of allowed states and it gives the atom an electron shell structure. After that orbit is full, the next energy level would have to be used. In 1922, Niels Bohr updated his model of the atom by assuming that certain numbers of electrons corresponded to stable “closed shells”. Bohr’s idea was that each discrete orbit could only hold a certain number of electrons. 
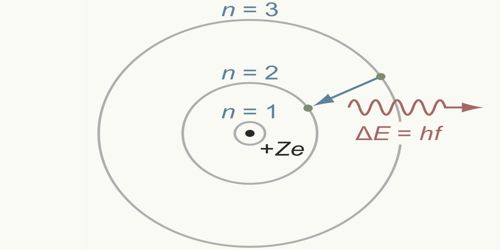
Heavier atoms, like carbon or oxygen, have more protons in the nucleus, and more electrons to cancel the charge. Subsequently, Bohr extended the model of hydrogen to give an approximate model for heavier atoms. The emission line spectrum of hydrogen tells us that atoms of that element emit photons with only certain specific frequencies ƒ and hence certain specific energies equal to E = hƒ, where h = Planck’s constant = 6.63 x 10 -34 J.s and f = frequency of the photon.Īlthough Bohr’s atomic model is designed specifically to explain the hydrogen atom, his theories apply generally to the structure of all atoms. Bohr’s theory was the first to successfully account for the discrete energy levels of this radiation as measured in the laboratory. The success of Bohr model is in explaining the spectral lines of atomic hydrogen and the Rydberg formula for the spectral emission lines. An atom can have an amount of internal energy equal to any one of these possible energy levels, but it cannot have an energy intermediate between two levels. Each of these stationary states is characterised by a specific amount of energy called its energy level. Therefore, each atom must be able to exist with only certain specific values of internal energy. During the emission of a photon, the internal energy of the atom changes by an amount equal to the energy of the photon. Planck’s hypothesis that energy is radiated and absorbed in discrete “quanta” (or energy packets) precisely matched the observed patterns of blackbody radiation and resolved the ultraviolet catastrophe.īased on this hypothesis, Bohr postulated that an atom emits or absorbs energy only in discrete quanta corresponding to absorption or radiation of a photon. Planck’s law is a pioneering result of modern physics and quantum theory. Planck’s quantum hypothesis ( Planck’s law) is named after a German theoretical physicist Max Planck, who proposed it in 1900. 
The Bohr model adopted Planck’s quantum hypothesis and he proposed a model in which the electrons of an atom were assumed to orbit the nucleus but could only do so in a finite set of orbits.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
